Public Health - Projects
Central Asia One Health National Actions Plans
VetEffect was awarded the project One Health National Actions Plans for Kazakhstan, Kyrgyz Republic, Tajikistan, Turkmenistan, and Uzbekistan by the Worldbank
The objective of the project is to formulate One Health National Actions Plans for pandemic preparedness, zoonoses, Antimicrobial Resistance (AMR) and food safety, for inclusion in the Central Asia One Health Regional Framework for Action (FfA). Recommendations will be based on an assessment of current activities, investment and needs, and agreed upon with Government counterparts in the Agriculture, Environment and Health sectors.

Kazakhstan, Kyrgyz Republic, Tajikistan, Turkmenistan, and Uzbekistan
VetEffect was awarded the project One Health Risk Assessment: Emerging and Re-emerging Infectious Disease Prioritization, Mapping, and Risk Assessment for Kazakhstan, Kyrgyz Republic, Tajikistan, Turkmenistan, and Uzbekistan by the Worldbank. The objective of this assignment is to develop list of priority emerging or re-emerging infectious diseases; suitability or risk maps of the top priority diseases of different epitopes and a a risk assessment report on the top priority diseases of different epi-types requiring a OH approach.
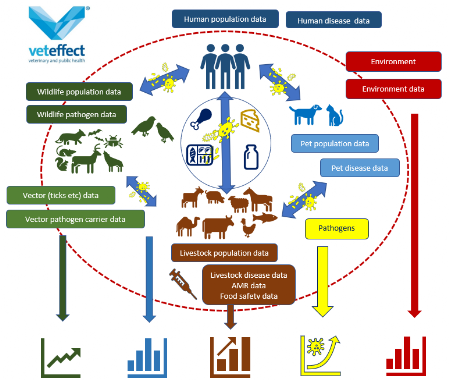
As part of the Mapping Climate Sensitive Zoonotic Diseases in Bangladesh - project by the WorldBank, VetEffecT aims to identify high-risk areas, while enhancing national capacity for prioritization methodologies and risk assessment by teaching national experts our approach to this project.

As part of the Biosecurity practices project, biosecure sampling of livestock and sample submission of hazardous animal infections will be strengthened and institutionalized from field to national reference laboratory. VetEffecT set up an e-learning environment to be used by international trainers to train and provide post-training consultancy, and by regional trainers for training of their national field veterinary staff and laboratory staff.
VetEffecT is conducting an AMR (antimicrobial resistance) project with Epiconcept (Fr) for the WHO to develop guidance for low- and middle-income countries on monitoring progress in addressing antimicrobial resistance (AMR) in a pragmatic and sustainable way. This should include the creative use of existing and emerging data sets.

VetEffecT participates in a One Health JPIAMR research network ARCH on AMR (antimicrobial resistance). The ARCH Network uniquely brings together multisectoral specialists and networks in the field of animal and human surveillance to bridge the gap between surveillance data and antibiotic stewardship in both compartments. Surveillance is essential to all aspects of the clinical management of antimicrobial resistance. It provides necessary information to develop empiric therapy guidelines, antibiotic formularies, and stewardship programmes. However, the value of surveillance as a critical component of antimicrobial stewardship is not fully established and the majority of the guidance documents focuses either on laboratory surveillance or antibiotic guidelines.

The European Centre for Disease Prevention and Control (ECDC) has established a large body of training materials for various functions around disease prevention and control. Produced by various authors, in different Member States, under different stages of an evolving strategy for capacity building through training, the library of training materials has developed organically, rather than systematically, in absence of a central standard or framework.

The study was carried out for the Worldbank. Consultancy Services for Strengthening Veterinary Institutional Capacity for Strategic Planning, Building Capacity for Economic Analysis of Zoonotic Diseases and Development of an inter-institutional ‘One Health’ Communications Strategy.
The purpose was to build Government capacity through the application of analytical tools and processes that provide guidance for the improvement of planning and decision-making related to animal health services and public health systems. The project follows OIE PVS Report, Gap Analysis Report and the OIE Strategic Report.
The project title is: “Organisation and implementation of training activities to strengthen EU law enforcement in Sanitary and Phytosanitary (SPS) fields in EU Member States under the “Better Training for Safer Food” initiative” (EAHC/2012/BTSF/08). It focuses on supporting Member State competent authorities to enforce EU rules on food and feed safety, animal health and welfare rules and plant health. Particular emphasis is placed on helping the Member States to develop the knowledge and capabilities needed to rectify issues disclosed during FVO missions. The overall objective of the proposed activities is to promote a systematic approach to the enforcement of EU SPS regulations by the competent authorities of participating Member States. A harmonised, risk-based and objective approach to enforcement enables competent authority resources to be employed efficiently so as to achieve uniform levels of compliance among food business operators across the EU.
The project aims at supporting the development of major functions of the SFDA deemed necessary for the execution of the mandate of the Authority in the areas of quality and safety of food and feed. The project is divided in several components, focused on capacity building of the SFDA head office, development of the strategy, and provision of technical assistance for the modernisation of the Border Inspection Posts (BIPs), for the modernisation of the food control laboratories in the country, and development of reference laboratory functions, set up of monitoring programmes for imported and locally produced food and feed, modernisation of analytical methods for microbiological and chemicals hazards, set up and improvement of the local inspection units, and IT and data management.

The project assisted Turkey in the implementation of EU policy regulations regarding the quality of dairy products that are incorporated in decision 92/46/EEC on health rules for the production and placing on the market of raw milk.

This study provides scenario's for origin labelling of products and the predicted economic consequences. The purpose was to evaluate scenario's for origin labelling of milk and milk products, to better inform consumer on the products' origin, yet minimising administrative burden and the principles of the internal market. The study was performed for the European parliament and Council regarding the mandatory indication of country of origin for types of meat other than beef, poultry, pig and sheep and goat meat, milk and milk used as an ingredient in dairy products, as a results of the Regulation (EU) No 1169/2011.

EDES aims to secure the flow of food products of animal and plant origin towards the EU or at regional level, in particular by making small-scale growers in Africa, Caribbean and Pacific countries key actors within the supply chain. COLEACP (Europe-ACP Liaison Committee), in cooperation with a consortium of European food safety agencies, has been entrusted by the European Union with the implementation of the EDES Programme. VetEffecT implements strengthening of veterinary and food laboratories including staff training and equipment procurement, and provides training of government staff of competent authorities and of private food business operators to be in conformity with EU and WTO-SPS regulations such as OIE, Codex Alimentarius, and IPPC .
The aim of the project is to execute an economic assessment of the impact of zoonotic diseases in Kazakhstan, Kyrgyzstan, Tajikistan and Uzbekistan in the mid-term and long-term context (10 years) and provision of evidential base for evaluation and development of additional program interventions and investments to mitigate these risks
